The magnitude and consequences of the COVID-19 pandemic will be all too clear to everybody. While writing this article, the number of people infected worldwide has topped 5 million and the number of deaths is over 300,000. In the United States alone, 100,000 people have died from the disease. It is natural for us to wonder what we can do about it. After all, this is a medical problem and so, like other medical problems, it should not be beyond humanity to devise ways of preventing the virus spreading and for curing people who are unfortunate enough to become infected. As has been widely stated, an effective vaccine would be a major step forward and efforts are underway to produce one. But vaccines are not necessarily easy to make. Here we should remind ourselves of the last major viral pandemic, the spread of HIV-1. A vaccine would also be an effective way of dealing with that problem. But people have been working on an effective HIV-1 vaccine for decades and nothing has come of it yet. So, while we wait and hope for a vaccine for treating COVID-19, we also need to consider other approaches. Here again we can take heart from thinking about the AIDS epidemic. A viral disease for which there was initially no cure, gradually became something we could control through the use of antiviral drugs. And it is in the area of pharmacology that many of our major hopes lie at the moment. It is the role of pharmacologists to come up with new drugs for treating diseases. So if, like me, you are a pharmacologist, then this is your moment. This is what you were born for. Come on pharmacologists—what have you got! The answer at the present time is not very much. But also not nothing. The first drugs that appear to make a dent in the course of SARS-CoV-2 infection (the name of the virus that produces the COVID-19 syndrome) are with us and the signs are that others may be on the way soon. So, what kinds of drugs are already being developed, how do they work and what might we expect in the future?
There are really two kinds of approaches to treating COVID-19 that might be amenable to drug development. The first of these is to produce drugs that attack the virus itself and prevent it infecting cells and replicating. Drugs such as these would be the best kinds of treatment. A second type of drug answers a slightly different problem. Let us assume that someone has an active infection; how can drugs target the consequences of this infection? SARS-CoV-2 infections can be fatal because they trigger adverse events that attack the body, leading to fatality. Drugs that target the molecules that enable these adverse events would also be extremely helpful and would save many lives.
To understand these two approaches we first need to understand the biology of the SARS-CoV-2 virus. Viruses are really the simplest forms of life. One might argue that they aren’t really alive at all in the conventional sense of the word, because in order to go through their normal life cycle, replicating to produce further viruses, they need the help of host cells. In effect they are tiny parasites that cannot live independently of their hosts. Viruses exhibit a range of preferences for infecting different species and different types of cells. There are over 200 types of viruses which show a preference for infecting humans rather than other animals, plants or even bacteria. The first of these to be discovered was Yellow Fever virus in 1901, and three to four new species are still being discovered every year. Some, like SARS-CoV (the virus that causes Severe Acute Respiratory Syndrome), MERS-CoV (the virus that causes Middle East Respiratory Syndrome) and SARS-CoV-2 (the virus that causes COVID-19) are extremely recent additions. Why pathogenic human viruses continue to appear isn’t completely clear, although it seems likely that viruses like SARS-CoV-2 originally existed in other species, such as bats in this case, and then mutated to produce new varieties that were able to utilize human hosts perhaps by passing through several other species first. The appearance of the HIV-1 virus in the latter part of the 20th century was another example of this kind of transmission, in that case initially from monkeys before jumping to humans. Coronaviruses were first identified in chickens in the 1930s and in humans in the 1960s. The viruses look like little balls with multiple spike-like structures sticking out of them and so resemble crowns, the feature that gave rise to their name. Initially, coronaviruses were thought to only produce mild symptoms in humans, such as the common cold. However, the appearance of the SARS-CoV, MERS-CoV and SARS-CoV-2 viruses has clearly demonstrated that newer members of the family can be much more pathogenic and dangerous. The SARS-CoV-2 virus has evolved to infect human cells (and possibly the cells of some other animals). Viruses normally consist of some genetic material, RNA or DNA, packaged into a protein coat, sometimes embedded in a lipid membrane. The purpose of the protein coat is to enable the viral nucleic acid to enter the host cell. Once inside, the viral genetic material takes over the host’s synthetic machinery, diverting it to the task of making more viruses which then lyse the cell and are free to infect further cells. Of the viruses that attack humans, SARS-CoV-2 is very large, being around 125 nanometers in diameter. SARS-CoV-2 viruses use a single-stranded RNA as their genetic material and have very large genomes consisting of 30,000 genetic bases. In fact, coronaviruses have the largest genomes of all RNA viruses. They are three times as big as HIV-1, for example.
How does SARS-CoV-2 infect humans and produce the particular symptoms associated with the COVID-19 syndrome? When somebody breaths in virus-laden droplets, SARS-CoV-2 enters the nose and throat. Many of the cells that line these regions express a protein receptor for the virus on their surface, called angiotensin-converting enzyme 2 (ACE2). ACE2 normally helps to regulate blood pressure, but the virus has evolved to be able to use it as a portal for entering target cells. It is the glycoprotein spikes that surround the virus and are made of a protein called “S-protein” that seek out and interact with ACE2 on the surface of target cells. SARS-CoV can also use the same protein as a receptor, but the interaction between SARS-CoV-2 and ACE2 is about 100 times stronger than that of SARS-CoV, providing one reason why it is highly infectious. Once the interaction between the virus S-protein and ACE2 has occurred, proteolytic enzymes on the cell’s surface—including furin or another enzyme called TMPRSS2—cleave the viral spike, exposing small peptides that help to fuse the viral membrane with the membrane of the host cell. Fusion allows the virus’ RNA to enter the host cell where it hijacks the host’s cellular machinery, diverting it from its normal role to producing viral RNA and proteins. These then get assembled into new virus particles which exit the host cell and can now attack other cells or leave the body and infect other people.

As the virus can only enter cells that express ACE2, it is important to understand exactly where in the body this receptor protein is expressed. The first cells to become infected are often in the throat and pharynx. The virus then moves along the windpipe (trachea) to attack the lungs and particularly the distant branches of the lung’s respiratory tree, which end in tiny air sacs called alveoli. These alveoli are lined by a single layer of cells that also highly express ACE2 receptors and so are easily infected by the virus. Infection of the lungs gives rise to many of the cardinal symptoms of SARS virus-related diseases. However, there are many other parts of the body that also express ACE2, including the cardiovascular system and brain. Some COVID-19 patients have strokes, seizures, confusion, and brain inflammation, indicating that the virus may also be able to directly attack the brain. The virus may also directly attack the lining of the heart and blood vessels, and so explain the cardiovascular symptoms displayed by many patients.
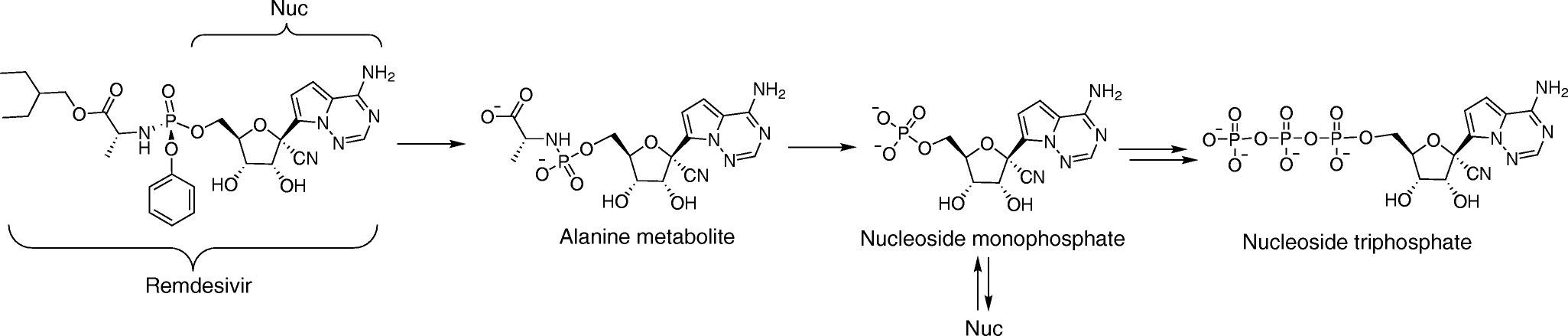
The different phases of the lifecycle of SARS-CoV-2 are well understood at this point and each one represents an opportunity for producing a drug that could be effective in blocking viral infection and replication. For example, one might imagine drugs that block the interaction between the virus S-protein and ACE2, drugs that interfere with the transcription and translation of viral genes and proteins, and drugs that interfere with the way that new virus particles are put together and exit cells. Experience with other viruses that are pathogenic in humans suggests that these approaches may also be possible with SARS-CoV-2. The era of antiviral drug development began with the first antiviral drug, idoxuridine, which was approved in June 1963. Now there are over 90 antiviral drugs that target 9 human infectious diseases including things like HIV-1 and influenza. Interestingly, some antiviral drugs have been approved for the treatment of more than one infectious disease, suggesting an underlying commonality in the processes through which different viruses replicate. This, of course, raises the hope that some existing antiviral drugs might be repurposed for treating COVID-19. Indeed, most of the initial drugs proposed for treating SARS-CoV-2 infection have been repurposed in this way. This is true, for example, in the case of remdesivir, a drug which has been widely discussed in the press as a potential COVID-19 treatment. Remdesivir is a novel small-molecule adenine nucleotide analogue antiviral drug that has shown efficacy against Ebola virus, another single-stranded RNA virus. Remdesivir also displays antiviral activity against other single-stranded RNA viruses, including filoviruses, pneumoviruses, paramyxoviruses, and the coronaviruses MERS-CoV and SARS-CoV. Remdesivir is what is called a “prodrug”, meaning that it has to be metabolized into its active form, another molecule called an adenine nucleotide triphosphate analogue that interferes with the activity of viral RNA-dependent RNA polymerase, the enzyme which transcribes the viral genome. Normally, the SARS-CoV-2 virus has a “proofreading” capacity, which allows it to detect potential mistakes in duplicating its RNA, but remdesivir can avoid this activity, leading to inhibition of viral RNA synthesis and termination of viral replication. It was found that patients with advanced COVID-19 and lung involvement who received remdesivir recovered faster than similar patients who received placebo, according to a preliminary data analysis from a randomized, controlled trial involving 1,063 patients, which began on February 21. Preliminary results have indicated that patients who received remdesivir had a 31% faster time to recovery than those who received placebo. Specifically, the median time to recovery was 11 days for patients treated with remdesivir compared with 15 days for those who received placebo. Results also suggested a survival benefit, with a mortality rate of 8.0% for the group receiving remdesivir versus 11.6% for the placebo group. Clearly, therefore, although remdesivir does not represent a way of completely curing the disease, it does appear to produce significant benefit to patients and is an interesting example of a drug that has been repurposed to treat COVID-19, a trend that will surely continue.
A second drug which has received a good deal of publicity is hydroxychloroquine. Hydroxychloroquine is president Trump’s drug of choice, and he is said to take it daily. As the Trump administration has pointed out, the fact that, as far as anybody knows, the president has not come down with COVID-19 is sufficient to prove its effectiveness as far as they are concerned. Like remdesivir, hydroxychloroquine is metabolized in the body to produce chloroquine, a drug with which humanity has had a great deal of experience. During World War II, the Japanese conquest of the Far East had cut off supplies of quinine, leaving the combatants desperate to find effective antimalarial drugs for their troops. Both the Germans and the Allies found that a synthetic drug called resochin, which had been made by the Bayer pharmaceutical company, was quite effective in controlling malaria but was initially considered too toxic for general use. Eventually, however, resochin (now renamed chloroquine) proved to be useful and, after many years of trials, became the most widely used antimalarial drug in the world. As one scientist put it, “The main story of chloroquine, 1934 to 1946, involves investigators in six countries on five continents and embraces its initial discovery, rejection, re-discovery, evaluation and acceptance.” Chloroquine proved to be a breakthrough drug, much more effective than any synthetic drug produced previously for the treatment of malaria. It was used throughout the world to treat many forms of malaria and also as a prophylactic measure. In some areas, it was even added to cooking salt to ensure everybody took it. Malaria isn’t caused by a virus but by a group of infectious organisms called Plasmodium spp. These are actually protozoans (single-cell organisms) rather than viruses but, like viruses, they lead a parasitic existence. Chloroquine kills malaria parasites by interfering with the way they dispose of the heme molecule in the red blood cells they infect, heme being toxic to Plasmodium spp. Hydroxychloroquine and chloroquine have subsequently proved to have other useful effects, including in the treatment of arthritis and lupus as well as general antiviral effects. Indeed, it is clear that these drugs can kill coronaviruses in “test tube” experiments carried out in a laboratory. An initial small clinical trial with hydroxychloroquine conducted in France in March appeared to show some beneficial effects in COVID-19 patients. Since that time however, further trials have proved discouraging. A large study in hospitalized COVID-19 patients in New York found that the drug had no impact on the risk of the most severe outcomes from the disease. Of the 1,446 patients admitted to the hospital from Mar 7 through April 8, 811 (58.9%) received HQ, with 45.8% of patients receiving treatment within 24 hours of presenting at the emergency department and 85.9% within 48 hours. Many of the patients who received HQ were classed as severely ill. No benefit was associated with HQ administration and this agrees with other trials that have also been carried out, and so enthusiasm for using the drug to treat COVID-19 patients appears to be poorly based in fact. Indeed, as I write this (May 26th, 2020), the World Health Organization has just announced that it was halting worldwide trials testing hydroxychloroquine for COVID-19. Last week, a study in the medical journal The Lancet reported that there were no benefits to treating coronavirus patients with hydroxychloroquine, and that taking it might even increase the number of deaths among those in hospital with the disease.
Other substances, mostly repurposed from previous applications, have shown activity against SARS-CoV-2 in the laboratory and are also being considered but have not yet been extensively tested in the clinic. These include compounds that inhibit the viral protease that is important for processing the proteins that constitute new viruses, compounds that block the interaction between viral S-proteins and ACE2, and compounds that inhibit proteases like TMPRSS2 that are involved in cleavage of the S-protein or molecules which, like remdesivir, interfere with the RNA-dependent RNA polymerase that transcribes the viral genome. These strategies all seem reasonable from a scientific point of view and could certainly ultimately turn out to produce something therapeutically useful.
Rather than considering these traditional targets, recent study published in the journal Nature took the approach of going “back to the drawing board” and trying to come up with entirely new targets for drugs to combat SARS-CoV-2. Gordon et al. (2020 Apr 30. doi: 10.1038/s41586-020-2286-9: online ahead of print) cloned the majority of the proteins that constitute the SARS-CoV-2 virus and then expressed them in human cells and measured which cellular proteins they interacted with. The authors argued that many of these interactions could be important in the normal processing of the virus and so drugs that inhibited them might have therapeutic effects. A large number of interactions were observed. However, after applying several other criteria, the authors whittled these down to a few targets that they deemed particularly interesting. After all the experimental dust had settled, the authors had a winner: sigma (σ) receptors! So, what are σ receptors and how might they be important? Back in the 1970s, there were many ongoing efforts to discover how many different types of receptors existed for opioid drugs. In addition to μ, κ and δ receptors, one categorization based on interactions with the drug N-allylnormetazocine (SKF10047) also came up with a class called σ, although in many respects this proposed receptor didn’t display properties that suggested it mediated the primary effects of opioid drugs. For one thing, the general opioid antagonist naloxone didn’t bind to the site and, moreover, in the case of many σ-selective drugs, the (+) isomer of the drug, which was usually devoid of opiate-like properties, proved to interact much more potently than the biologically active (-) isomers. Further studies using (+) pentazocine as a defining ligand showed that the σ binding site could not be defined as an opioid receptor but must have some other function. Indeed, the σ receptor proved to have affinity for a spectrum of chemicals that had been previously shown to exhibit a wide range of pharmacological effects. Moreover, a second binding site (σ2) was also defined and subsequently found to be identical with a previously known protein called TMEM97, which had been suggested as being a transport protein involved in cholesterol biosynthesis. In spite of a lot of work done over the years—including structural work, the preparation of knockout mice and many other important studies—nobody has ever pinned down exactly what the biological functions of σ receptors might be. There is a strong impression that they may be involved in modulating biochemical signaling pathways in cells that are activated by other receptors such as G protein-coupled receptors, but really nobody knows and σ receptors remain a biological mystery. Nevertheless, Gordon et al. demonstrated that several molecules that bind to σ receptors were powerful inhibitors of SARS-CoV-2 production in cultured cells, suggesting that they may be a novel therapeutic target in this case. Although these experiments are very interesting and suggestive, it should be pointed out that historically σ receptors have been “discovered” as being novel targets for most of the diseases that afflict humanity, including pain, amyotrophic lateral sclerosis, Alzheimer’s disease, Parkinson’s disease, retinal disease, stroke, cocaine and alcohol addiction and, of course, cancer. Unfortunately, σ-specific drugs have yet to be translated for any useful therapeutic advantage in human patients. Hence, whether σ receptors are really the answer to the SARS-CoV-2 problem awaits further testing, particularly in COVID-19 patients.
As indicated above, another important method for potentially intervening in COVID-19 is to find a way of ameliorating the destructive effects that result from viral infection. The viral receptor ACE2 is also expressed in some immune cells, including monocytes, macrophages and dendritic cells. SARS-CoV-2 infection of these cells results in their activation and secretion of IL-6 and other inflammatory cytokines. IL-6 has particularly powerful proinflammatory properties and has been implicated as a key mediator of many inflammatory syndromes. The receptor for IL-6 consists of an IL-R protein combined with a second protein named gp130. Soluble forms of IL-R can combine with circulating IL-6 and then this complex can associate with gp130 proteins, which are very widely expressed by many tissues including endothelial cells. The resulting IL-6-mediated signaling triggers the synthesis of many other inflammatory cytokines, producing a “cytokine storm” that can lead to vascular permeability and leakage, which participate in the pathophysiology of hypotension and pulmonary dysfunction in COVID-19. Cytokine storms have been shown to produce pathology in a variety of situations such as during CAR-T cell treatment for cancer, where they can produce very dangerous and ultimately fatal effects. This has led to the suggestion that cytokine storms may be controlled by treatments that neutralize circulating IL-6, such as the humanized monoclonal antibody tocilizumab which has been shown to be effective in reducing these problems during CAR-T cell therapy. Cytokine storms have been suggested as being triggered by SARS-CoV-2 infection and are thought to be responsible for many of the far-reaching effects of the virus in tissues that it doesn’t primarily infect, and so treatments with agents such as tocilizumab may be helpful, something that appears to be the case in some early clinical trials.
The arrival of the COVID-19 pandemic, as well as the government funding that goes with it, has meant that many scientists have converted their laboratory efforts to researching this problem, resulting in an enormous worldwide effort to produce novel drugs for combating the disease. Hopefully, some combination of new drugs and vaccines will reduce the SARS-CoV-2 problem to just another manageable infection. Nevertheless, the appearance of SARS-CoV, MERS-CoV and SARS-CoV-2 in rapid succession means that there are many lessons about preparedness to be learned before the next new pathogen turns up.
